Personalized, Multisession, Multichannel Transcranial Direct Current Stimulation in Medication-Refractory Focal Epilepsy: An Open-Label Study
Abstract
Purpose: Animal and proof-of-principle human studies suggest that cathodal transcranial direct current stimulation may suppress seizures in drug-resistant focal epilepsy. The present study tests the safety, tolerability, and effect size of repeated daily cathodal transcranial direct current stimulation in epilepsy have not been established, limiting development of clinically meaningful interventions.
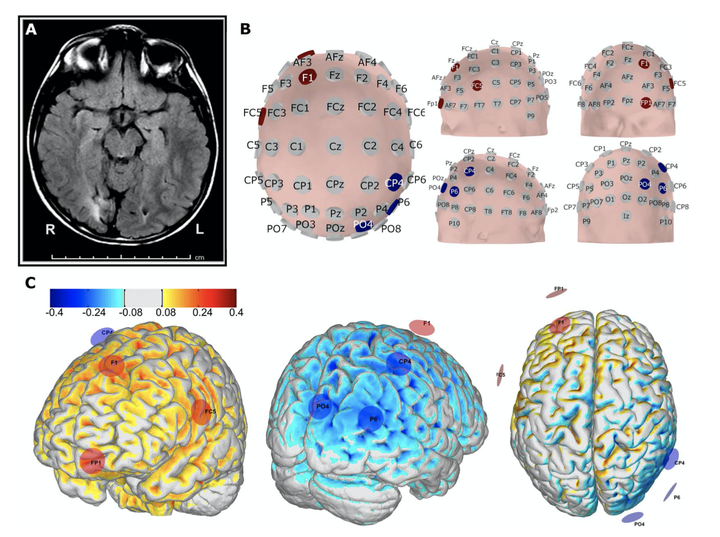
Methods: We conducted a 2-center, open-label study on 20 participants with medically refractory, focal epilepsy, aged 9 to 56 years (11 women and 9 children younger than18 years). Each participant underwent 10 sessions of 20 minutes of cathodal transcranial direct current stimulation over 2 weeks. Multielectrode montages were designed using a realistic head model-driven approach to conduct an inhibitory electric field to the target cortical seizure foci and surrounding cortex to suppress excitability and reduce seizure rates. Patients recorded daily seizures using a seizure diary 8 weeks prior, 2 weeks during, and 8 to 12 weeks after the stimulation period.
Results: The median seizure reduction was 44% relative to baseline and did not differ between adult and pediatric patients. Three patients experienced an increase in seizure frequency of >50% during the stimulation period; in one, a 36% increase in seizure frequency persisted through 12 weeks of follow-up. Otherwise, participants experienced only minor adverse events-the most common being scalp discomfort during transcranial direct current stimulation.
Conclusions: This pilot study supports the safety and efficacy of multifocal, personalized, multichannel, cathodal transcranial direct current stimulation for adult and pediatric patients with medication-refractory focal epilepsy, although identifies a possibility of seizure exacerbation in some. The data also provide insight into the effect size to inform the design of a randomized, sham-stimulation controlled trial.